BOSTON — Do you have questions about the coronavirus vaccine? Here’s what we know so far:
Before getting vaccinated
Will I have to pay for the vaccine?
The vaccine is being provided free of charge to all individuals by the federal government. Insurance companies are also committed to not charging any out-of-pocket fees or co-payments related to COVID-19 vaccine administration, and all health care provider sites that receive COVID-19 vaccine must agree to not charge patients any out-of-pocket fees or deny anyone vaccination services.
May undocumented immigrants receive the vaccine for free?
Yes. The vaccine itself is free for all Massachusetts residents. Health insurance (including Medicare and Medicaid) will cover the cost of administering the vaccine. For patients without health insurance, health care providers may request reimbursement from the federal government for the cost of administering vaccine to undocumented immigrants.
Will getting the vaccine adversely impact a patient’s immigration status?
No. The federal government has confirmed that it will not consider COVID-19 treatment (including a vaccine) as part of a determination of whether someone is a “public charge” or as it relates to the public benefit condition for certain individuals seeking an extension of stay or change of status, even if the vaccine is paid for by Medicaid or other federal funds.
Is a patient’s vaccination record protected from disclosure?
The Department of Public Health will maintain an electronic record of each patient in Massachusetts who receives the COVID-19 vaccine. The vaccine database is kept confidential like a patient’s medical record with her doctor.
Will the COVID-19 vaccine be mandatory?
The Department of Public Health is not mandating the COVID-19 vaccine. It is a voluntary program. The COVID-19 vaccine has been shown to be highly effective at preventing illness and it is an important tool in the fight against the pandemic.
Will I need to be tested for COVID-19 before getting the vaccine?
No, a COVID-19 test is not needed before getting the vaccine.
Should someone who is COVID-19-positive receive the vaccine?
Not if they are symptomatic. Vaccination of people who are known to have COVID-19 should be delayed until they no longer have symptoms.
Should people who have had COVID-19 be vaccinated?
Yes, people who have previously had COVID-19 should be vaccinated.
What are the benefits of getting a COVID-19 vaccine?
We understand that some people may be concerned about getting vaccinated now that COVID-19 vaccines are available in the United States. Please visit Benefits of Getting a COVID-19 Vaccine | CDC for a summary of the benefits of COVID-19 vaccination based on what we currently know.
How will vaccines work against new COVID-19 variants, like the one that appeared in the United Kingdom?
It is normal for viruses to change as they spread, and for new variants to appear. Scientists are working to learn more about new COVID-19 variants to understand how easily they might spread, and whether the vaccines we already have will protect people against them. Right now, we don’t have any evidence that the new COVID-19 variants can make people sicker or increase risk of death. However, as time goes on and scientists learn more, we will have more detailed information.
Why would a vaccine be needed if we can do other things, like social distancing and wearing masks, to prevent the virus that causes COVID-19 from spreading?
Stopping a pandemic requires using all the tools available. Vaccines work with your immune system so your body will be ready to fight the virus if you are exposed. Other steps, like covering your mouth and nose with a mask and staying at least 6 feet away from others, help reduce your chance of being exposed to the virus or spreading it to others. Together, COVID-19 vaccination and following CDC’s recommendations to protect yourself and others will offer the best protection from COVID-19. (source: Frequently Asked Questions about COVID-19 Vaccination | CDC as of 12/21/20)
What is an Emergency Use Authorization?
The Pfizer and Moderna vaccines have received Emergency Use Authorization (EUA) from the federal Food and Drug Administration. Learn more about what this means – and other information about the vaccines – in their EUA fact sheets for recipients and caregivers. These factsheets can be found at: Pfizer-BioNTech COVID-19 Vaccine EUA Fact Sheet for Recipients and Caregivers (fda.gov) and Moderna COVID-19 Vaccine EUA Fact Sheet for Recipients and Caregivers (fda.gov)
Who is authorized to administer vaccinations?
In addition to licensed health care providers, like doctors and nurses and pharmacists, according to the federal CARES Act, vaccinations may be administered by pharmacy interns, certified pharmacy techs, paramedics, and certified medical assistants, under the direct supervision of a primary care provider. When available, all health care institutions including hospitals, community health centers, and private medical offices will have access to the COVID-19 vaccine to vaccinate their patients. Pharmacies including large retail outlets such as CVS, Walgreens, Osco, and Walmart will have access to the vaccine and will be able to vaccinate their customers. Vaccine will likely also be made available to local health departments for vaccinating their local residents at public clinics.
This COVID-19 Vaccinators chart lists all of the different groups of individuals who can possess and administer COVID-19 vaccines.
During your appointment
What can I expect at my appointment to get vaccinated for COVID-19?
Please visit What to Expect at Your Appointment to Get Vaccinated for COVID-19 | CDC for tips on what to expect when you get vaccinated, what information your provider will give you, and resources you can use to monitor your health after you are vaccinated.
Do I need to wear a mask when I receive a COVID-19 vaccine?
Yes. CDC recommends that during the pandemic people wear a mask that covers their nose and mouth when in contact with others outside your household, when in healthcare facilities, and when receiving any vaccine, including a COVID-19 vaccine. Anyone who has trouble breathing or is unable to remove a mask without assistance should not wear a mask. For more information, visit considerations for wearing masks. (source: Frequently Asked Questions about COVID-19 Vaccination | CDC as of 12/21/20)
Can I choose which vaccine I receive?
No. Recipients will receive the vaccine offered to them when they attend a vaccination clinic. Both vaccines are highly effective and safe.
How long do I have to wait between the first and second dose of the COVID-19 vaccine?
The Pfizer vaccine requires two doses to be given 21 days apart. The Moderna vaccine requires two doses to be given 28 days apart.
Will I receive anything to show proof of vaccination?
You should receive a vaccination card or printout that tells you what COVID-19 vaccine you received, the date you received it, and where you received it. You should receive a paper or electronic version of a fact sheet that tells you more about the specific COVID-19 vaccine you are being offered. Each authorized COVID-19 vaccine has its own fact sheet that contains information to help you understand the risks and benefits of receiving that specific vaccine. The factsheets can be found at Pfizer-BioNTech COVID-19 Vaccine EUA Fact Sheet for Recipients and Caregivers (fda.gov) and Moderna COVID-19 Vaccine EUA Fact Sheet for Recipients and Caregivers (fda.gov)
After getting vaccinated
What should I do if I experience symptoms after receiving a COVID-19 vaccine, such as fever and body aches?
If you have any significant pain or discomfort, talk to your healthcare provider, who may recommend over-the-counter medicine, such as ibuprofen or acetaminophen. To reduce pain and discomfort where you got the shot apply a clean, cool, wet washcloth over the area, and use or exercise your arm. To reduce discomfort from fever, drink plenty of fluids and dress lightly. In most cases, discomfort from fever or pain is normal, but contact your healthcare provider if: the redness or tenderness where you got the shot increases after 24 hours your side effects are worrying you or do not seem to be going away after a few days
How do I report if I have any side effects after getting the COVID-19 vaccine?
V-safe is a smartphone-based tool that uses text messaging and web surveys to provide personalized health check-ins after you receive a COVID-19 vaccination. Through v-safe, you can quickly tell CDC if you have any side effects after getting the COVID-19 vaccine. Depending on your answers, someone from CDC may call to check on you and get more information. And v-safe will remind you to get your second COVID-19 vaccine dose if you need one. To sign up for v-safe, please visit V-safe After Vaccination Health Checker | CDC.
How long after getting the COVID-19 vaccine does it take to be effective?
It typically takes a few weeks for the body to build immunity after vaccination. That means it’s possible a person could be infected with the virus that causes COVID-19 just before or just after vaccination and get sick. This is because the vaccine has not had enough time to provide protection.
How long does protection from the COVID-19 vaccines last?
We do not have data yet to say for how long the COVID-19 vaccines will provide protection from getting sick even if you do get COVID-19. Experts continue to conduct more studies about whether the vaccines also keep people from spreading COVID-19.
Does immunity after getting COVID-19 last longer than protection from COVID-19 vaccines?
The protection someone gains from having an infection (called natural immunity) varies depending on the disease, and it varies from person to person. Since this virus is new, we don’t know how long natural immunity might last. Some early evidence—based on some people— seems to suggest that natural immunity may not last very long. Regarding vaccination, we won’t know how long immunity lasts until we have a vaccine and more data on how well it works. Both natural immunity and vaccine-induced immunity are important aspects of COVID-19 that experts are trying to learn more about, and CDC will keep the public informed as new evidence becomes available. (source: Frequently Asked Questions about COVID-19 Vaccination | CDC as of 12/21/20)
How effective is one dose of a COVID-19 vaccine compared to two doses?
The COVID-19 vaccines were not studied for use as a single dose. People should get both doses of the vaccine to be fully vaccinated in order to be effective.
Do I have to follow the Travel Order if I have received a COVID-19 vaccine?
Yes. At this time, vaccinated individuals must continue to comply with the Governor’s Travel Order and related testing and quarantine requirements. It is important for everyone to continue using all the public health measures available to help stop this pandemic. Cover your mouth and nose with a mask when around others, stay at least 6 feet away from others, avoid crowds, and wash your hands often.
Do I need to wear a mask and avoid close contact with others if I have received two doses of the vaccine?
Yes. While experts learn more about the protection that COVID-19 vaccines provide under real-life conditions, it will be important for everyone to continue using all the tools available to us to help stop this pandemic, like covering your mouth and nose with a mask, washing hands often, and staying at least 6 feet away from others. Together, COVID-19 vaccination and following CDC’s recommendations for how to protect yourself and others will offer the best protection from getting and spreading COVID-19. Experts need to understand more about the protection that COVID-19 vaccines provide before deciding to change recommendations on steps everyone should take to slow the spread of the virus that causes COVID-19. Other factors, including how many people get vaccinated and how the virus is spreading in communities, will also affect this decision. (source: Frequently Asked Questions about COVID-19 Vaccination | CDC as of 12/21/20)
When can I stop wearing a mask and avoiding close contact with others after I have been vaccinated?
There is not enough information currently available to say if or when CDC will stop recommending that people wear masks and avoid close contact with others to help prevent the spread of the virus that causes COVID-19. Experts need to understand more about the protection that COVID-19 vaccines provide before making that decision. Other factors, including how many people get vaccinated and how the virus is spreading in communities, will also affect this decision. (source: Frequently Asked Questions about COVID-19 Vaccination | CDC as of 12/21/20)
Accessing the vaccine
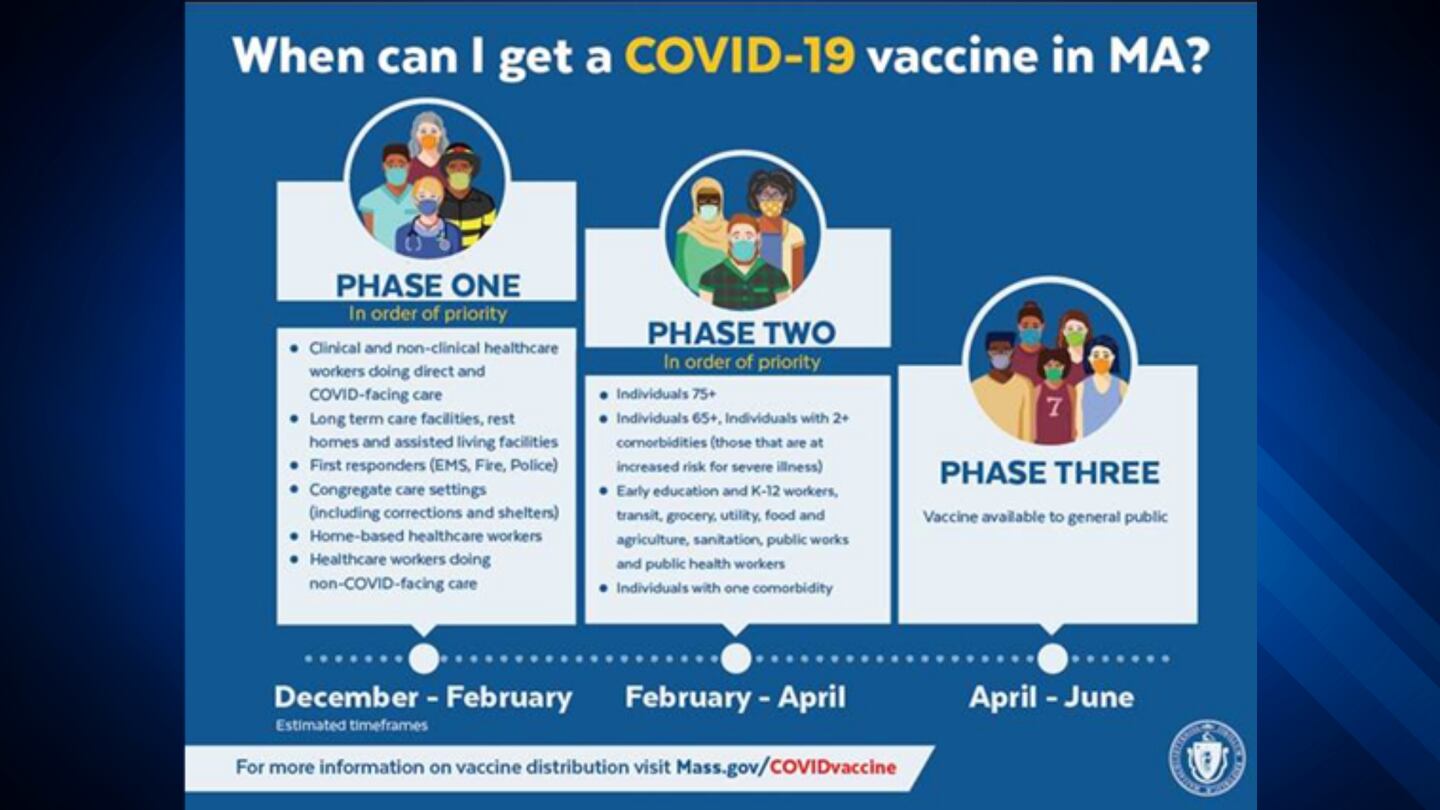
Who will get vaccine first?
Vaccine prioritization will occur in phases. Learn more about the distribution timeline here.
When can I get the COVID-19 vaccine?
Please visit When can I get the COVID-19 vaccine? to learn where you will fit into the COVID-19 vaccine distribution timeline.
How will I know that it is my turn to get the COVID-19 vaccine?
Most people in the Commonwealth are scheduled to receive the vaccine in Phase 3 of the Massachusetts Covid-19 Vaccination Plan. As the state moves through the prioritization phases, information on where, when and how to get the vaccine will be announced publicly and posted on COVID-19 Vaccine in Massachusetts | Mass.gov. At this time, the Department of Public Health is not scheduling appointments directly. In Phase 1 of the Massachusetts COVID-19 Vaccination Plan, you will be contacted by the health care facility, nursing home or other entity that is tasked with vaccinating COVID-facing health care workers or long-term care residents. If you are a first responder, you are eligible to get vaccinated0starting January 11. For more information, visit When can I get the COVID-19 vaccine? | Mass.gov In Phase 2 of the Massachusetts COVID-19 Vaccination Plan, the Department of Public Health expects to work through treating clinicians/health systems, health insurers (including MassHealth), and patient advocacy groups to reach out to individuals with co-morbidities to encourage them to receive COVID-19 vaccination. Individuals in the various critical-worker professional groups will likely be reached via their employers and unions to inform them of the importance of COVID-19 vaccination.
How were decisions made about who to prioritize in the Massachusetts COVID-19 Vaccination Plan?
Across the country, experts have focused on how to distribute limited quantities of COVID-19 vaccines in a fair, ethical, and transparent way with the goal of reducing serious illness and death, preserving the health care infrastructure, preserving critical functions of society, advancing health equity, and addressing the needs of the most vulnerable first. The National Academies of Sciences, Engineering, and Medicine (NASEM) and CDC’s Advisory Committee on Immunization Practices (ACIP) have made recommendations regarding prioritization of groups. In Massachusetts, a COVID-19 Vaccine Advisory Group made recommendations to the Secretary of Health and Human Services and the Governor about how to prioritize distribution of the vaccine in the Commonwealth, carefully considering the work of NASEM and ACIP. The Advisory Group included medical professionals, public health experts, community leaders, elected officials, and infectious disease specialists. The three primary goals of the Advisory Group’s recommendations were to: preserve the health care system; limit severe morbidity and mortality; and promote equity. For more information about the COVID-19 distribution timeline in Massachusetts, visit When can I get the COVID-19 vaccine? | Mass.gov.
What will be done to prioritize those disproportionately impacted by COVID-19?
In Phases 2 and 3, Massachusetts will set aside 20 percent of the vaccine to providers in communities that have been the most affected by COVID-19. These communities will be decided based on US Census data and COVID-19 cases per capita on the city and town level.
What co-morbidities will be prioritized during Phase 2 of the state’s COVID-19 Vaccine Plan?
In phase 2, Massachusetts will prioritize people with underlying medical conditions for which they are at increased risk of severe illness from COVID-19, as defined by the CDC. At this time, this does not include people with conditions for which they might be at an increased risk for severe illness.
Will people who live in another state or country part-time (e.g. students, retirees, people with dual citizenship) be able to get the COVID-19 vaccine in Massachusetts?
The Massachusetts COVID-19 Vaccination program is intended for individuals who live, work or study in the Commonwealth. At this time, people will get vaccinated either by their employer, primary care provider, pharmacy or public clinic depending on the phase of the Vaccination Plan for which they are eligible.
If I received the first dose of vaccine in another state, may I get the second in Massachusetts?
Yes. You would get your second dose here either by your employer, primary care provider, pharmacy, or public clinic depending on the phase of vaccination for which you are eligible. Please be sure to keep the vaccination card you were given at the time of the first dose.
Vaccine safety
How do we know if the vaccine is safe?
It’s important to know that vaccines go through more testing than any other pharmaceuticals. Before any vaccine is made available, it must go through rigorous development and testing. Manufacturing is critical — every dose must consistently be high quality. Additionally, extensive testing in clinical trials is conducted to prove safety. First, small groups of people receive the trial vaccine. Next, vaccine is given to people with particular characteristics (e.g., age and physical health). Then, vaccine is given to tens of thousands of people and tested for effectiveness and safety. After that, the data is reviewed by the FDA which approves the vaccine, and by an independent board, CDC’s Advisory Committee on Immunization Practices (ACIP) which will make its recommendations for use. These bodies are the final safeguards for the public ensuring any vaccine is both safe and effective. Please visit Ensuring the Safety of COVID-19 Vaccines in the United States | CDC for more information.
Do the COVID-19 vaccines have any side effects?
It is possible that some people may have side effects after being vaccinated, which are normal signs that your body is building protection. These side effects may affect your ability to do daily activities, but they should go away in a few days. For the Pfizer vaccine, the most common side effects are pain at the injection site, tiredness, headache, muscle pain, chills, joint pain, and fever. More people experienced these side effects after the second Pfizer dose than after the first dose, so it is important to expect that there may be some side effects after either dose, but even more so after the second. For the Moderna vaccine, the most common side effects are pain, tenderness, swelling and redness at the injection site, as well as fatigue, headache, muscle pain, joint pain, chills, nausea and vomiting, and fever.
Can a COVID-19 vaccine make me sick with COVID-19?
No. The vaccines do not contain the live virus that causes COVID-19. This means that a COVID-19 vaccine cannot make you sick with COVID-19. (source: Facts about COVID-19 Vaccines (cdc.gov) as of 1/20/21)
Should someone with a history of allergies get the COVID-19 vaccine?
You should not get the Pfizer or Moderna COVID-19 vaccines if you have a history of severe allergic reaction (e.g., anaphylaxis) to any ingredient in the vaccine. A history of a severe allergic reaction due to any other cause is considered a precaution and should be discussed with your health care provider before receiving the vaccine. Although there is a small chance that the COVID-19 vaccines could cause a severe allergic reaction, this would usually happen within a few minutes to one hour after getting the vaccine. If you have a history of allergic reactions, your vaccination provider may ask you to stay at the place where you received your vaccine for monitoring afterwards.
What are the ingredients in the vaccine?
Both Pfizer and Moderna have fact sheets for recipients and caregivers that list the ingredients of each vaccine. Those factsheets can be found at: Pfizer-BioNTech COVID-19 Vaccine EUA Fact Sheet for Recipients and Caregivers (fda.gov) (page 2), and Moderna COVID-19 Vaccine EUA Fact Sheet for Recipients and Caregivers (fda.gov) (page 2).
I would like to have a baby one day. Is it safe for me to get a COVID-19 vaccine?
Yes. People who want to get pregnant in the future may receive the COVID-19 vaccine. COVID-19 vaccines are being studied carefully now and will continue to be studied for many years, similar to other vaccines. Based on what we know right now, experts believe that COVID-19 vaccines are safe for someone who is trying to become pregnant in the short or long term. Here’s why: The COVID-19 vaccine, like other vaccines, works by teaching our bodies to develop antibodies that fight against the virus that causes COVID-19, to prevent future illness. There is no evidence right now that antibodies formed from COVID-19 vaccination will cause any problems with pregnancy, including the development of the placenta. In fact, there is no evidence that fertility problems are a side effect of ANY vaccine. People who are trying to become pregnant now or who plan to try in the future may receive the COVID-19 vaccine when it becomes available to them. (source: Facts about COVID-19 Vaccines (cdc.gov) as of 1/20/21)
Should someone who is pregnant or breastfeeding get a COVID-19 vaccine?
Currently there are no data on the safety of COVID-19 vaccines in people who are pregnant. COVID-19 infection during pregnancy can increase the risk of severe illness and might result in an increased risk of outcomes like preterm birth. Getting vaccinated is a personal choice for people who are pregnant. There are no data on the safety of COVID-19 vaccines in people who are breastfeeding or on the breastfed baby. People who are breastfeeding may choose to be vaccinated. The American College of Obstetricians and Gynecologists recommend that COVID-19 vaccines be offered to breastfeeding individuals when they meet prioritization criteria for receiving the vaccine. If you are pregnant or breastfeeding and have questions around getting vaccinated, a discussion with your healthcare provider might help you make an informed decision.
Are the COVID-19 vaccines safe for children?
At this time, the Pfizer vaccine is authorized for people ages 16 and older, and the Moderna vaccine is authorized for people ages 18 and older. Younger children and adolescents should not receive COVID-19 vaccination at this time.
Who were the vaccines tested on?
The safety of the Pfizer COVID-19 vaccine was evaluated in people 16 years of age and older in two clinical studies conducted in the United States, Europe, Turkey, South Africa, and South America. Overall, 50.6% of participants were male and 49.4% were female, 83.1% were White, 9.1% were Black or African American, 28.0% were Hispanic or Latino, 4.3% were Asian, and 0.5% were American Indian or Alaska Native. (Pfizer-BioNTech COVID-19 Vaccine EUA Fact Sheet for HCP revised 01-06-2021) The safety of the Moderna COVID-19 vaccine was evaluated in people 18 years of age and older in the United States. Overall, 52.7% of participants were male, 47.3% were female, 79.2% were White, 10.2% were Black or African American, 20.5% were Hispanic or Latino, 4.6% were Asian, 0.8% were American Indian or Alaska Native, 0.2% were Native Hawaiian or Pacific Islander, 2.1% were Other, and 2.1% were Multiracial. (Moderna COVID-19 Vaccine EUA Fact Sheet for Healthcare Providers) There have been no studies in people who are pregnant or breastfeeding.
Will a COVID-19 vaccine change my DNA?
No. COVID-19 mRNA vaccines do not change or interact with your DNA in any way. Messenger RNA vaccines—also called mRNA vaccines—are the first COVID-19 vaccines approved in the United States. mRNA vaccines teach our immune system how to fight against a specific virus. COVID-19 mRNA vaccines work with the body’s natural defenses to safely develop immunity to disease. In order to do its job, the mRNA from a COVID-19 vaccine doesn’t need to go inside the nucleus of the cell, which is where our DNA is kept. This means the mRNA never interacts with our DNA in any way, and has no way to change it. At the end of the process, our bodies have learned how to protect against future infection. That immune response and making antibodies is what protects us from getting infected if the real virus enters our bodies. (source: Facts about COVID-19 Vaccines (cdc.gov) as of 1/20/21)
More information:
Visit these frequently updated Centers for Disease Control and Prevention (CDC) web pages on COVID-19 vaccination:
- Benefits of Getting a COVID-19 Vaccine
- How COVID-19 Vaccines Work
- Myths and Misconceptions about COVID-19 Vaccines
- Frequently Asked Questions about COVID-19 Vaccination (newly expanded)
Download the free Boston 25 News app for up-to-the-minute push alerts
>> Complete local and national coronavirus coverage here
RESOURCES:
- Complete local and national coronavirus coverage here
- Follow us on Facebook and Twitter | Watch Boston 25 NOW